AAV.CPP.16, an adeno-associated virus variant, can effectively pass through the blood-brain barrier of the central nervous system, and deliver drugs to the brain and spinal cord, which proves malignant glioma could be effectively treated in laboratory mice and cynomolgus monkeys. Recently, scholars from the Neuropsychiatric Hospital at Renmin Hospital of Wuhan University (Hubei General Hospital) published ground-breaking research in Nature Biomedical Engineering. The result provides an important new tool for gene therapy of central nervous system diseases, which signals significant progress in gene therapy of human central nervous system diseases, further consolidating the hospital’s leading position in the research of nervous system diseases in China.
On October 10, a research paper called Variants of the adeno-associated virus serotype 9 with enhanced penetration of the blood-brain barrier in rodents and primates was published online in the internationally renowned journal Nature Biomedical Engineering (impact factor 29.234). Using screening, the study found that AAV.CPP.16 can effectively pass through the blood-brain barrier of the central nervous system, deliver drugs to the brain and spinal cord, and fully prove the therapeutic efficacy of the carrier in a mouse model with malignant glioma.
Wang Jun, the co-first author of this paper, and the associate chief physician of neurosurgery at Renmin Hospital of Wuhan University, has long been engaged in basic research and clinical diagnosis and treatment of the central nervous system, dealing with tumors and vascular diseases. In 2018, he participated in Professor Bei Fengfeng’s team to study at the Department of Neurosurgery, Brigham and at the Women’s Hospital of Harvard Medical School.

Wang Jun explained that gene therapy refers to the introduction of exogenous normal genes into target cells, to achieve treatment purposes by correcting or compensating for diseases caused by defects or abnormal genes. It possesses a broad application prospect for the treatment of tumors, vascular diseases, hereditary diseases and other fields. Adeno-associated virus (AAV) is a small virus that is currently the main platform for gene therapy delivery in corpore. Through a variety of strategies such as gene replacement, gene silencing, gene editing or expression of exogenous antibodies, one-time treatment of disease can be achieved. In May 2019, the US FDA approved the first gene therapy drug Zolgensma using an intravenous AAV9 carrier for the treatment of children under 2 years of age with spinal muscular atrophy.
In the study of gene therapy, researchers have been able to confirm that the low delivery efficiency of the central nervous system (CNS) is an important bottleneck, deterring the development of AAV9-mediated CNS gene therapy. Wang Feng explained that the blood-brain barrier is a layer of tightly packed cells arranged on the blood vessel wall of the brain, which can prevent toxins, pathogens and macromolecular substances from entering the brain. It is an important barrier to protect the central nervous system evolved by the body. Unfortunately, it is also what majorly hinders gene therapy drugs from entering the central nervous system. Therefore, how to break through the blood-brain barrier and deliver the target gene is the main problem of gene therapy of the nervous system.
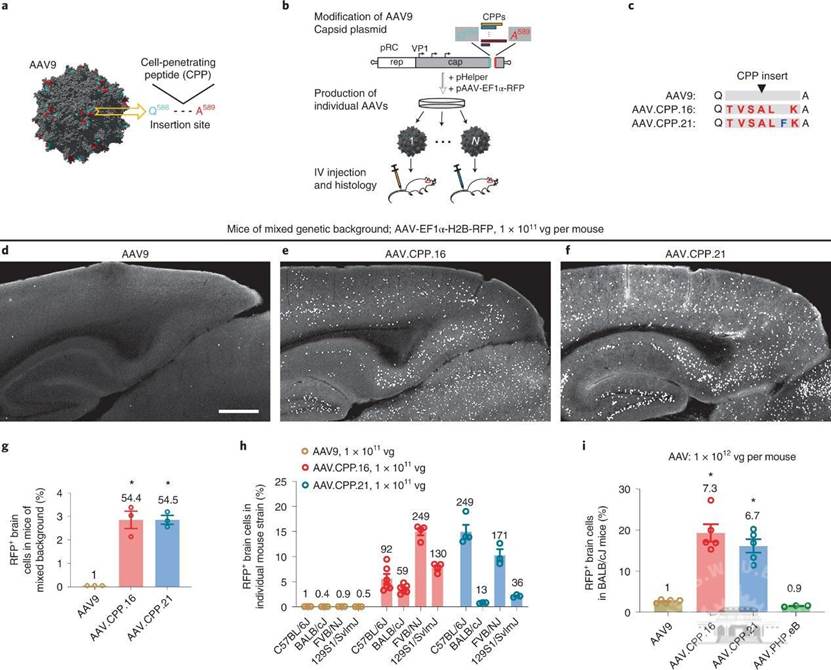
During the study of variants of the adeno-associated virus serotype 9 with enhanced penetration of the blood-brain barrier in rodents and primates, Wang Jun and other researchers used rationally designed methods to modify the cell-penetrating peptides (CCPs) on the capsids of wild-type AAV serotype 9 and screened out AAV variant AAV.CPP.16 with high blood-brain barrier penetration. This AAV variant can quickly cross the blood-brain barrier, and even through intravenous injection, to form efficient, widespread infection and expression in the CNS.
In the animal experiments, AAV.CPP.16 carrying the target gene was injected into different strains of mice, and juvenile and adult cynomolgus monkey animal models. It was confirmed that compared with other AAV variants, the transmission efficiency of AAV.CPP.16 across the blood-brain barrier was significantly improved, and the target gene could be efficiently and safely expressed in the CNS of the primate cynomolgus monkey. This is a breakthrough in the global research of AAV variants, and is also a big step forward in the gene therapy of central nervous system diseases.
Meanwhile, Wang Jun et al. designed AAV CPP.16-aPD-L1-HA carrying vectored single-chain variable fragment antibody expressing PD-L1 (programmed death ligand 1) and hemagglutinin (HA) protein gene, which was injected intravenously into fluorescent GL261 glioma model mice. By about 26 days, all the mice in the blank control group had died, while six of the eight mice in the gene therapy group had survived for more than 100 days.
The surviving mice were reseeded with brain tumors and given a new control group. The control mice also all died within four weeks, while the gene-treated mice remained healthily alive until day 200, the equivalent of 60 to 70 human years. Later tests of brain tissue from surviving mice found no identifiable tumor cells but only scar-like tissues in the area where the tumor was planted, meaning that this group of mice had been cured of glioma once and for all. Mice in another glioblastoma model, GBM1694, also achieved nearly 50% longer survival than the control group.
These findings indicate that gene drugs using AAV CPP.16 as a vector have long-lasting and effective anti-tumor effects, and these findings will be further applied in clinical practice in the future.


The corresponding author of the paper, Professor Bei Fengfeng, has been researching synaptic injury and gene-drug development for many years. He said the findings bring the successful conversion of AAV to deliver drugs across the human blood-brain barrier one step closer. It also suggests that AAV can be used as an effective systemic drug delivery tool against glioma or other central nervous system diseases in need.
The Neurosurgery Department of Renmin Neuropsychiatric Hospital of Wuhan University is a national key clinical specialty. It is a strong team with a remarkable research group, including Professor Chen Qianxue, Liu Renzhong, Chen Zhibiao, Jian Zhihong and other well-known experts in the field of nervous system malignant tumors and cerebrovascular diseases. Members of the team have published more than 200 articles in Nature Biomedical Engineering, Advanced Functional Materials, Bioactive Materials, Small, Theranostics, Journal of Nanobiotechnology, Clinical and Translational Medicine and other internationally renowned journals and worked as chief editor, chief translator and co-editor on more than ten monographs.
It is reported that the People’s Hospital of Wuhan University prioritizes the protection of people’s health, and attaches great importance to scientific and technological innovation. Centering on organs and systems, the hospital maximizes the integration of “ace + ace” medical resources, and combines medicine and surgery departments as well as preponderant departments. Five branch hospitals have been established, including a cardiovascular clinic, a neuropsychiatric clinic, an eye, ear, nose and throat clinic, an internal medicine clinic and a urology and kidney clinic. While providing more high-quality, efficient, accurate and convenient integrated health and medical services, they constantly stimulate the vitality of scientific and technological innovation through mechanism innovation, and continue to contribute wisdom and strength to tackle clinical problems that urgently need to be solved.
Rewritten by Kang Jiayi & Jiang Hanjun
Edited by Zhang Yuhua, Sylvia, Xi Bingqing