Professor Chen Caiyou and his team from the College of Chemistry and Molecular Sciences at Wuhan University have achieved a breakthrough in organic chemistry, with their latest research on electrochemical copper-catalyzed C(sp3)-C(sp2) electrophilic cross-coupling published online in the Journal of the American Chemical Society.
The study, Expanding Ullmann Coupling to Cross-Coupling: Electrochemical Copper-Catalyzed Electrophilic Cross-Coupling of Alkyl and Aryl Halides, presents a novel approach to the construction of C(sp3)-C(sp2) bonds.
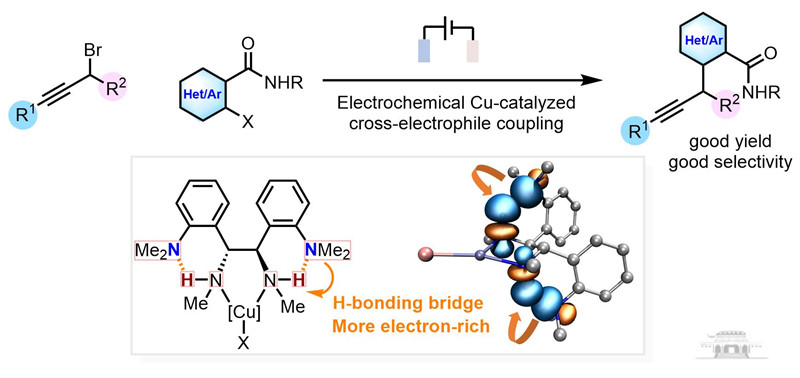
This groundbreaking research expands the classical Ullmann coupling, traditionally reliant on organometallic reagents or active methylene substrates, to a more versatile cross-coupling of alkyl and aryl halides.
The team has designed a new bidentate ligand with ortho-dimethylamino groups, enhancing electron-donating capabilities through intramolecular hydrogen-bond-promoted hyperconjugation.
The team achieved high selectivity in cross-coupling, suppressing side reactions through rapid halogen transfer and the stabilization of organic copper intermediates. The reaction is compatible with various propargyl bromides and (hetero)aryl halides, operating under mild conditions and exhibiting excellent functional group tolerance.
Mechanistic studies indicate that the reaction proceeds via a radical pathway, with in situ electrochemical mass spectrometry capturing a trivalent copper intermediate. This research opens a new electrochemical pathway for copper-catalyzed C(sp3)-C(sp2) bond construction, showcasing significant potential for synthetic applications.